Acceleration of bone healing: Identification of a therapeutic target in a pre-clinical model
Uma Sankar
Indiana University School of Medicine, USA
: J Forensic Toxicol Pharmacol
Abstract
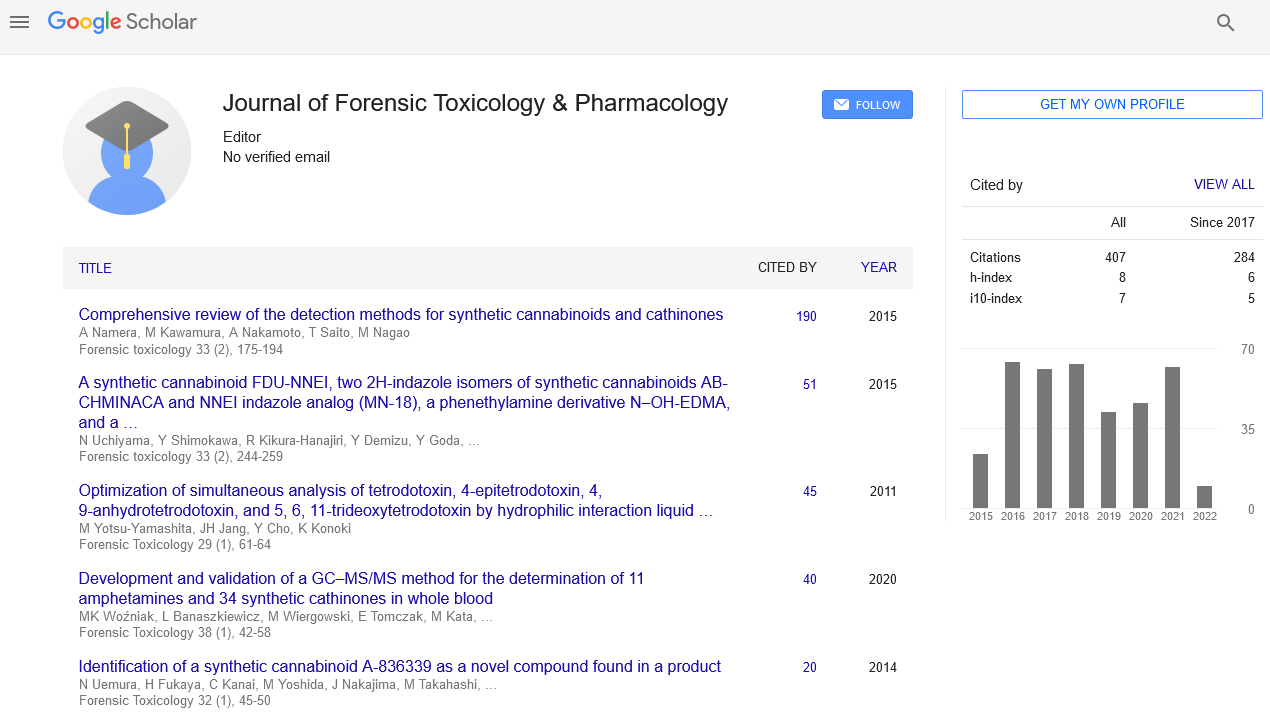
Fractures associated with osteoporosis and acute trauma result in significant medical costs, loss of productivity and patient quality of life. Currently, there are no effective pharmacological treatments that promote efficient healing of bone fractures. Ca2+/calmodulin (CaM)-dependent protein kinase kinase 2 (CaMKK2) has roles in the anabolic and catabolic pathways of bone remodeling. Its pharmacological inhibition with STO-609 protects from post-menopausal osteoporosis and reverses age-associated bone loss. In this study, we hypothesized that targeting CaMKK2 will accelerate fracture healing. To this end, unilateral femoral fractures were generated in 10 week old male C57BL6 mice. Tri-weekly intraperitoneal injections of saline (n=30) or STO-609 (n=30; 10 μmol/kg body weight) were administered for 4 weeks post-fracture. Fractured calluses were analyzed at days 3, 7, 14 and 28 days by micro-computed tomography (micro-CT), immunohistochemistry and qPCR to assess healing. During normal fracture healing in mice, hypertrophic chondrocytes appear at the callus around day 14, produce vascular endothelial growth factor (VEGF) which elicits migration of mesenchymal stem cells (MSCs). Treatment with STO- 609 results in a marked elevation in hypertrophic chondrocytes and VEGF as well as a dramatic influx of MSCs in the callus by day 7. By day 14, these calluses possess significantly higher levels of osteocalcin and calcified matrix compared to controls. Micro-CT analyses reveal that STO-609-treated calluses possess significantly more bony-callus area by 2 weeks and mature bone by 4 weeks post-fracture. Thus, STO-609-treated mice possess more mature and stronger secondary bone in their calluses indicating faster repair of the fracture. Toxicology analyses indicate no alteration in blood or hepatic biochemistry following STO-609 treatment. Altogether, our observations reveal that CaMKK2 inhibition using its selective pharmacological inhibitor STO-609 results in the acceleration of key early cellular and molecular mechanisms involved in fracture healing such that healing is accelerated by a whole week.
Biography
Uma Sankar investigates the precise role of Ca2+/calmodulin-dependent protein kinase (CaMK) signaling in bone remodeling and maintenance, with a key emphasis on translational studies involving CaMK kinase 2 (CaMKK2) pharmacological inhibition as a bone anabolic therapeutic strategy in the prevention and reversal of post-menopausal, age-as well as treatment-induced osteoporosis, fracture healing and osteoarthritis.
Email: usankar@iupui.edu
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi