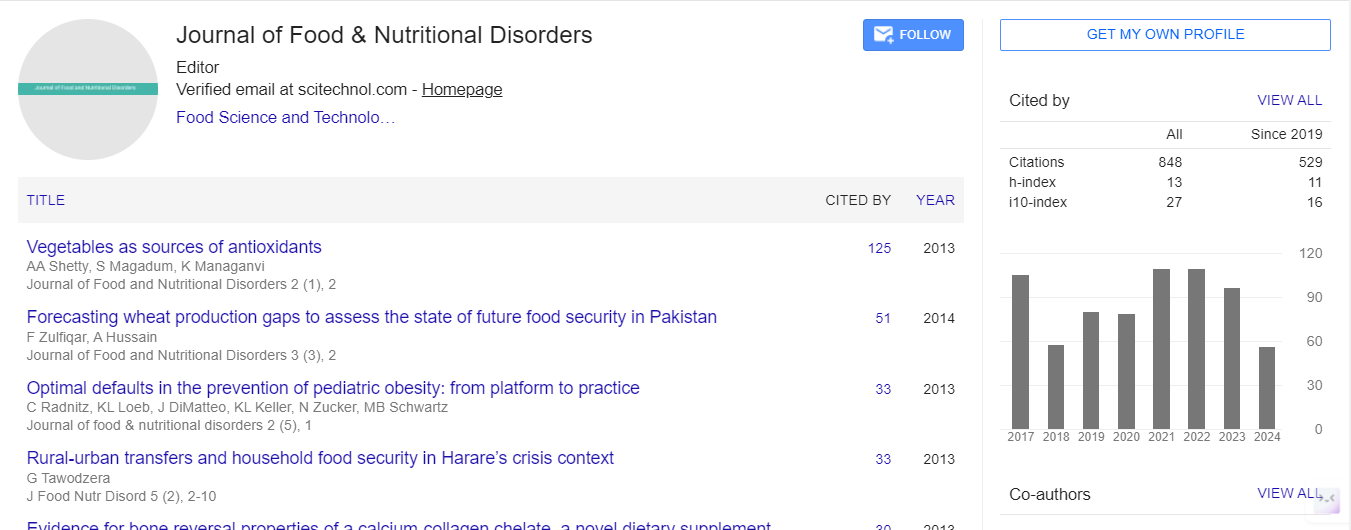
Opinion Article, J Food Nutr Disor Vol: 13 Issue: 5
Integrating Multi-Omics Approaches to Identify Potential Therapeutic Targets in Alzheimer’s through Nutritional Science
Oliver Reynolds*
1Department of Bioinformatics, ETH Zurich University, Zurich, Switzerland
*Corresponding Author: Oliver Reynolds,
Department of Bioinformatics, ETH Zurich University, Zurich, Switzerland
E-mail: reynoldsoilver2@gmail.com
Received date: 24 September, 2024, Manuscript No. JFND-24-151331;
Editor assigned date: 26 September, 2024, PreQC No. JFND-24-151331 (PQ);
Reviewed date: 11 October, 2024, QC No. JFND-24-151331;
Revised date: 21 October, 2024, Manuscript No JFND-24-151331 (R);
Published date: 28 October, 2024, DOI: 10.4172/2324-9323.1000431
Citation: Reynolds O (2024) Integrating Multi-Omics Approaches to Identify Potential Therapeutic Targets in Alzheimer’s through Nutritional Science. J Food Nutr Disor 13:5.
Description
The Alzheimer's disease (AD) poses a significant global health challenge, marked by progressive neurodegeneration, cognitive decline and eventual loss of independence. Recent advancements in research have highlighted the complexity of AD, involving difficult molecular pathways that necessitate a comprehensive understanding, particularly from a nutritional and food science perspective, to identify potential therapeutic targets. Integrating multi-omics approaches-encompassing genomics, transcriptomics, proteomics and metabolomics-has emerged as a powerful framework to clarify the mechanisms underlying AD and uncover novel opportunities for dietary interventions.
Multi-omics refers to the simultaneous study of multiple biological layers. In the context of food science, genomics investigates genetic predispositions to dietary metabolism; transcriptomics examines how dietary components affect gene expression; proteomics analyzes proteins that interact with nutrients; and metabolomics focuses on metabolic byproducts resulting from dietary intake. By integrating these diverse data types, researchers can achieve a integrated understanding of the biological processes influenced by diet in relation to AD, moving beyond the limitations of single-omics studies that often fail to recognize the interactions between food components and various molecular levels. One of the key advantages of multi-omics integration is its capacity to reveal complex interactions that contribute to AD pathology, particularly regarding dietary factors. For instance, genetic studies have identified several risk alleles, such as those in the APOE gene, which are known to modulate lipid metabolism a important aspect influenced by dietary fats and oils.
Coupled with transcriptomic data, researchers can examine how specific dietary components affect the expression of genes involved in these metabolic pathways, leading to a better understanding of how nutrition influences cognitive decline and neurodegeneration. Moreover, the interaction between various omics layers can clarify how dietary habits modify gene expression and metabolite profiles, potentially influencing Alzheimer's risk. By integrating these environmental factors, including dietary patterns, researchers can develop comprehensive models that account for the multifactorial nature of AD, prepare for personalized dietary recommendations aimed at reducing disease progression.
The integration of multi-omics data is particularly advantageous for biomarker discovery, which is essential for early diagnosis and monitoring of Alzheimer’s. Identifying specific dietary-related proteins or metabolites that correlate with disease progression could facilitate the development of quantitative assays for clinical applications. For instance, integrated analyses of metabolomics and proteomics may reveal unique metabolic signatures associated with dietary influences on neurodegeneration, serving as potential biomarkers for therapeutic targeting or patient stratification in clinical trials. Recent studies utilizing multi-omics platforms have identified biomarkers signaling the onset of Alzheimer's that are influenced by nutritional factors. For example, alterations in lipid metabolism and inflammatory profiles in the brains of individuals with AD have been linked to dietary patterns, providing insights into the pathogenic mechanisms and highlighting specific, diet-modifiable pathways that could be targeted therapeutically.
Identifying novel therapeutic targets through a nutritional lens is another critical application of multi-omics integration in Alzheimer’s research. By understanding the roles of specific dietary proteins, fats, and other nutrients in the progression of Alzheimer’s, researchers can design food-based interventions aimed at modulating these pathways effectively. The discovery of dysregulated signaling pathways influenced by nutrition, such as those involving neurotransmitter systems or neuroinflammatory responses, can guide the formulation of dietary strategies for restoring normal function. Moreover, multiomics approaches can assist in identifying existing food compounds with unforeseen efficacy in treating Alzheimer’s. By analyzing the molecular profiles of AD patients alongside dietary intake data, researchers can explore strategies for dietary repositioning, where refined dietary interventions might serve new therapeutic roles. Integrating multi-omics approaches in Alzheimer’s research represents a transformative shift towards personalized nutrition. As technological advancements continue, the ability to analyze extensive datasets will enhance our understanding of the multifactorial nature of the disease, allowing for targeted dietary interventions tailored to specific patient subgroups. Nonetheless, this integrated approach requires robust datasharing initiatives and collaborative research efforts across food science and clinical research to realize its full potential.
To conclude, integrating multi-omics offers an unprecedented opportunity to explore the complex biological networks related to Alzheimer's disease from a food science perspective. By revealing novel therapeutic targets and potential biomarkers connected to dietary factors, this strategy not only enhances our comprehension of AD pathology but also opens the door to innovative dietary interventions tailored to the unique molecular characteristics of individuals affected by this challenging condition. As ongoing research progresses, it holds the promise of refining therapeutic approaches available for Alzheimer's disease through targeted nutritional strategies.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi