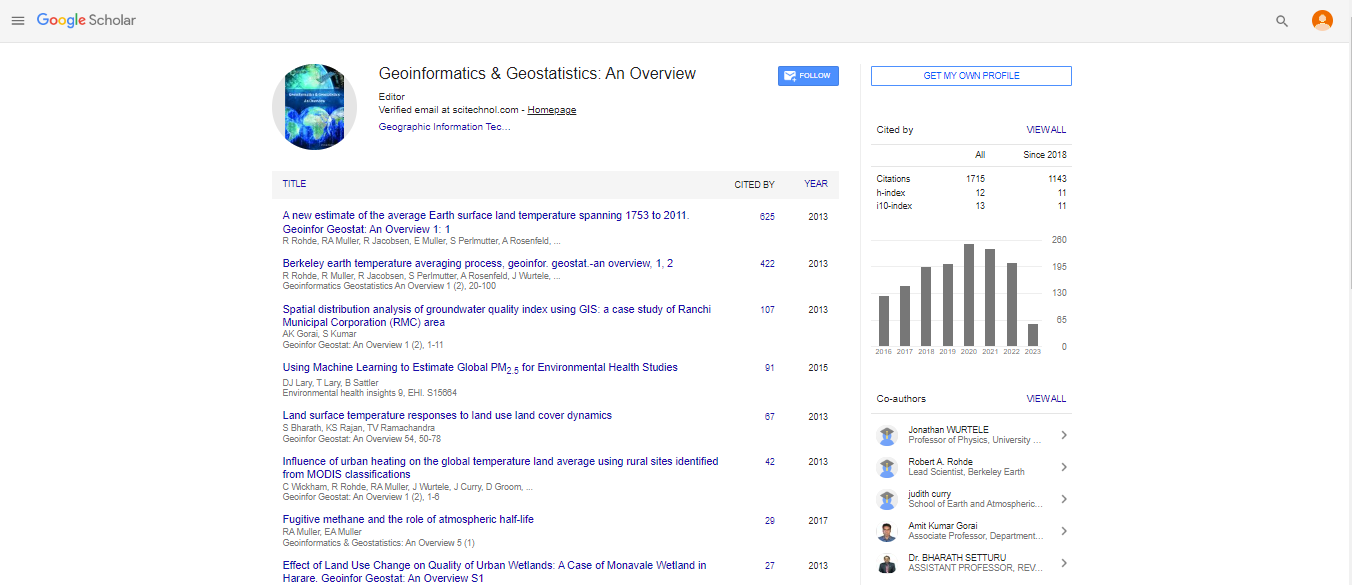
Expert Review, Geoinfor Geostat An Overview Vol: 12 Issue: 5
Chemistry Analysis for the Origin of Life in Protoplanetary Disks
Xiaoming Li*
1Department of Geography and Planning, University of Toronto, Toronto, Canada
*Corresponding Author: Xiaoming Li,
Department of Geography and Planning, University of Toronto, Toronto, Canada
E-mail: xli835ca@yahoo.ca
Received date: 13 September, 2024, Manuscript No. GIGS-24-147967;
Editor assigned date: 16 September, 2024, PreQC No. GIGS-24-147967 (PQ);
Reviewed date: 30 September, 2024, QC No. GIGS-24-147967;
Revised date: 07 October, 2024, Manuscript No. GIGS-24-147967 (R);
Published date: 14 October, 2024, DOI: 10.4172/2327-4581.1000411.
Citation: Xiaoming Li (2024) Chemistry Analysis for the Origin of Life in Protoplanetary Disks. Geoinfor Geostat: An Overview 12:5.
Abstract
The primary purpose of this review is to present chemistry analysis. In this theoretical study analyzing the origin of life, the main argument is that life on Earth may not have originated on Earth but rather in the protoplanetary disk at the time of the solar system's formation. This analysis focuses on the discoveries and problems encountered by scientists in the limited field of origin of life research and proposes possible solutions. These discussions include the chemical characteristics of protoplanetary disks and their complex systems that appear more conducive to life's origin. The water paradox for the origin of life on Earth may be resolved on protoplanetary disks; the chiral preference for amino acids on Earth may have originated on protoplanetary disks. The molecular composition of protoplanetary disks may be more conducive to explaining the origin of life; the large number of organic molecules observed in cosmic dust may have originated on protoplanetary disks. The ultraviolet resistance of life on Earth may have originated in the protoplanetary disk. Life may have originated on protoplanetary disks through archaeological chemical analysis of the Morrison and other meteorites. Analyze literature on the last universal common ancestor to explore the possibility that life originated on protoplanetary disks. Finally, model the possible origin of life on protoplanetary disks and explain the potential origin of life on protoplanetary disks. Discussions of the theoretical basis for the origin of life on protoplanetary disks are limited to theoretical analysis. Hopefully, these theoretical discussions will provide scientists engaged in the origin of life research with a new vision of the origin of life research. These discussions will contribute to the future development of the origin of life research.
Keywords: The origin of life; Protoplanetary disk; Biological
evolution; The Universe
Introduction
There are three fundamental questions about origins: The origin of the Universe, the Origin of Life (OoL) and the origin of mind and consciousness. These questions have been debated for centuries and are now sub-topics of serious scientific research [1]. The origin of life on Earth from the Earth itself, also known as the "internal Earth origin" or "biogenesis," is one of the dominant theories about the origin of life in the life sciences. The theory holds that life on Earth evolved gradually from inorganic matter through natural processes and has nothing to do with extraterrestrial life or exotic organisms— however, the "primordial soup" Obalin-Holden hypothesis", the 'Miller-Urey experiment'. The 'RNA world hypothesis and its 'hydrothermal vent theory' are all mainstream theories that life on Earth originated on Earth [2-5].
Another theory proposed by a few scientists is the Panspermia Theory, which suggests that life did not originate on Earth alone but is spread throughout the Universe. Such life forms can be microorganisms or simpler organic molecules [6]. Panspermia suggests that microbes or seeds of life may have attached themselves to cosmic dust, comets, meteorites, or other celestial bodies and spread through space. These celestial bodies may have carried life from one planet to another, including Earth [7].
These theories of the origin of life have made significant contributions to the origin of life. Scientists have elaborated on the possible causes of the origin of life in different ways and from various perspectives. In 1859, Darwin published the origin of species [8]. Darwin's tree of biological evolution had only one trunk because it represented the scientific consensus that all life forms originated from a common ancestor, a theory known as the theory of a single origin or the theory of a single origin. Scientists have a growing consensus that the "exogenous transport" of organic matter was combined with the "endogenous synthesis" of chemical evolution. These two processes complement each other and together, they complete the process of the origin of life. However, scientists are far from completing the jigsaw puzzle of the origin of life.
Today, most of the debate among scientists about the origin of life focuses on the following four points:
• The geological environment
• The source of food/starting molecules
• The source of energy
• The delineation between the RNA world and the first part of metabolism [9].
However, there is little scientific consensus among scientists on the origin of life. Life on Earth arose from self-organizing inanimate matter away from thermodynamic equilibrium involving networks of autocatalytic reactions and protoplasm formation. So, where else in our planetary system, besides Earth, does such a move away from a state of thermodynamic equilibrium exist? It has been found that the protoplanetary disk of our solar system is also in this state, far from thermodynamic equilibrium [10]. Can we open our eyes to the possibility that life originated in the protoplanetary disk? The early Earth environment is very different from the protoplanetary disk environment, but the state of being far from thermodynamic equilibrium is the same. Are there many other factors in the protoplanetary disk favoring the origin of life that we do not know about? Below, it will discuss the following aspects of scientists' research on the origin of life and its challenges and explore the possibility of considering solutions to these questions from protoplanetary disks.
Discussion
A Protoplanetary Disk (PROD) is usually disk-shaped. It is a flat structure of gas and dust surrounding a young star, the site of planet formation. Its disk-like structure is due to the conservation of angular momentum, which causes matter to form a flat disk as it rotates. This disk cools as it forms and dust particles collide and collect with each other, eventually forming planets and other small bodies [11].
The protoplanetary disk is a source of roots for the tree of the origin of life and biological chemical evolution
About 4.6 billion years ago, a shock wave from the eruption of one or more supernovae triggered the collapse of a localized region of this molecular cloud that eventually formed the Sun and its planetary system. Matter flowed through the disk to the young star at the center, a process known as accretion, forming an accretion disk. In some cases, the material in the stellar cloud disk helps the star grow and form planets through physical processes within the disk, which are called protoplanetary disks [11]. The Earth is thought to have been born 4.54 billion years ago and protoplanetary disks last about a million years to ten million years. These nearly ten million years may have been enough time for inorganic molecules to evolve into simple organic molecules, complex organic molecules and finally, the birth of cellular life. In a broader analysis, many organic molecules may have been formed during the stellar cloud phase, creating more favorable conditions for the subsequent synthesis of chemical molecules, the chemical evolution of protoplanetary disks and the eventual formation of life.
Chemical characteristics of protoplanetary disks
The protoplanetary disks contain a large number of CHNOPS elements, which are the main constituents of living matter, enriched with various "metals" including, but not limited to, alkali metals (Na,K), alkaline-earth metals (Mg,Ca), transition metals, posttransition metals (Zn,Al) and lanthanide elements (La-Lu), etc. [12]. The role of transition metals is emphasized here. The multivalent nature of transition metals makes them ideal catalytic centers. Transition metal ions are rich in coordination chemistry. They can form stable coordination complexes with various ligands such as water molecules, amino acid side chains and metal-organic cofactors. This diversity enables enzymes to precisely regulate the chemical environment of the active site precisely, thereby improving catalytic efficiency. The electronic structure of transition metals allows them to stabilize high-energy intermediates during catalysis and lower the reaction's activation energy by altering the electron distribution. As a result, many critical metabolic responses (e.g. nitrogen fixation, respiration, etc.) can be carried out efficiently.
The redox gradient formation is considered a critical factor in primitive chemical processes and may have provided essential conditions for the origin of life. Protoplanetary disks can form these redox gradients at different scales through various physicochemical processes that drive electrons to move between reduction and oxidation sites. Photochemical reactions: Solar radiation, incredibly ultraviolet light, can initiate photochemical reactions, such as photooxidation, in surface material. These reactions lead to changes in the oxidation state in specific regions, forming redox gradients in protoplanetary disks [13].
The Dynamic Kinetic Stability (DKS), enthalpy (H) and entropy (S) on protoplanetary disks may be very different from those on Earth.
These factors should be more favorable for the birth of life on protoplanetary disks. Dynamic kinetic stability is a fundamental concept in biological evolution. Unlike traditional thermodynamic stability, DKS is concerned with the stability of a system in a nonequilibrium state. Biological molecules or systems with high DKS are more likely to survive the evolutionary process because they can efficiently self-replicate and maintain structural stability [14]. Protoplanetary disks became progressively better and the catalysis of early enzymes may have depended heavily on enthalpy effects, which allowed these enzymes to maintain efficient biochemical reactions as the protoplanetary disks gradually cooled [15]. Entropy is an essential indicator for understanding spontaneous processes and equilibrium states in living systems. According to the second law of thermodynamics, the total entropy of an isolated system always tends to increase. In biological systems, an increase in entropy is usually associated with the diffusion and dispersion of energy. Conversely, decreases in entropy are related to the formation of ordered structures, such as protein folding or cell membrane formation. The evolutionary process involves the gradual complexification of an organism, which appears to be the process of entropy reduction. However, this complexification is achieved by consuming energy and increasing environmental entropy [16]. This increase in ecological entropy may only be realized on protoplanetary disks, especially by the first life.
The above analysis demonstrates the dynamic equilibrium from high to low temperatures required to produce primitive life and its celestial dissipation system that favours the origin of life. If we further study protoplanetary disks that are more compatible with the above conditions, it may provide us with new ideas for exploring the origin of life.
The Earth may not be able to accomplish the complex systems required for the origin of life and only protoplanetary disks may be able to do so
The above discussion of the origin of life does highlight the difficulty of envisioning the origin of life in a single environment. This challenge arises from the multiple complex chemical processes and environmental conditions required for the origin of life, which may not have existed chemically simultaneously or concurrently on the early Earth. Thus, understanding the origin of life requires exploring prebiotic processes and gaining insight into the environmental properties of the early Earth-multiple Environmental Requirements [17]. The origin of life may have involved various stages, each requiring specific environmental conditions. For example, the synthesis of certain organic molecules may have required a high temperature environment, while other molecules may have required a more relaxed atmosphere for stabilization. Thus, a single environment may not be able to accommodate all the necessary chemical processes simultaneously the complexity of environmental conditions [18]. The chemical complexity of life requires the synergistic effect of multiple environmental conditions. For example, the presence of water, the concentration of electrolytes, temperature variations, the action of light and the presence of metal ions are all critical factors required for the formation of life. Environmental conditions may fulfill these needs at different times and places [19]. These factors determine which environments can support the gradual increase in chemical complexity that ultimately led to the origin of life.
The various environmental requirements needed for the origin of life may not have been met in the same place on Earth. However, due to the complex processes of chemical evolution and primitive life, it was challenging for simple to complex organic molecules and primitive life cells to be met in the same place on early Earth. This condition creates the environmental paradox of the origin of life on Earth. How do we solve this problem? Can we turn our attention to the protoplanetary disk? All of the complex systems mentioned above for the origin of life could have existed on protoplanetary disks. Studying the possibility that life originated in the protoplanetary disk may be a way to solve the environmental paradox of the origin of life on Earth.
Protoplanetary disk theory could solve the "water problem" of nucleic acid polymerization
During the formation of nucleic acids (such as RNA and DNA), phosphodiester bonds between nucleotides are generated by a condensation-dehydration reaction. This reaction requires the removal of water molecules to form a stable polymer chain. However, water serves as both a solvent for the reaction and also inhibits the reaction because the presence of water drives the reaction in the opposite direction (i.e., , hydrolysis) [20]. The environmental requirements of the condensation reaction are highly unfavorable in an aqueous environment, as water promotes the hydrolysis of already-formed phosphodiester bonds, thus making the stabilization of nucleic acids challenging. This problem is particularly acute in the chemistry of early life, as the environments in which life originated were often water-rich and achieving stable polymerization of macromolecules in such environments is challenging. The phosphodiester bonds in RNA molecules are readily hydrolyzed in aqueous environments, especially at high temperatures or under alkaline conditions. Even if RNA is successfully formed under certain conditions, it may rapidly depolymerize in regular aqueous environments, challenging its stability as a molecule storing genetic information. Water molecules attack the phosphodiester bond, causing it to break. This spontaneous hydrolysis makes the long-term stability of RNA in water problematic, further exacerbating the water paradox in the origin of life [21]. Thus, we see the reason for the formation of the water paradox. Water provides the solvent environment needed for life and facilitates specific chemical reactions. However, water also destroys life molecules through a reverse reaction (hydrolysis). This contradiction leads to the paradox of the "water problem." Environmental constraints on the origin of life in the early Earth require that the chemical origin of life must occur in an environment that favors molecular polymerization while avoiding the excessive effects of hydrolysis [22]. How to successfully synthesize biomolecules in water has become a pressing challenge for scientists. Thus, those who believe life originated on Earth can expand their new thinking because of the "water paradox."
If we consider the "water paradox," we may be able to turn our attention to the idea that life on Earth originated on a protoplanetary disk. If life formed on the protoplanetary disk as replicable protocellular life and this life was splashed on Earth by a meteorite, then life could have reproduced and evolved on Earth. Thus, the problem of the "water paradox" is solved. That is, there may be no water molecules in the protoplanetary disk to break the phosphodiester bonds of the RNA hydrolysis polymers and thus, RNA and DNA could be preserved in the protoplanetary disk without any problems. Of course, it is also possible that the phosphodiester bonds between nucleotides could be polymerized into RNA or DNA by condensationdehydration reactions, which may also provide a favorable environment for the next step of cellular metabolism.
Life originated in the protoplanetary disk in terms of the composition of its chemical elements
The protoplanetary disk contains many Carbon, Hydrogen, Nitrogen, Oxygen, Phosphorus and Sulfur (CHNOPS) chemical elements (the main components of living matter) and many transition metals. Life would not have existed without the Transition Metals (TMs), at least not in the form we know. Transition metals in the first row of the periodic table are ubiquitous in all living species; 30% of proteins are associated with transition metals and 40% of enzymatic reactions are processed by metalloenzymes [23]. Therefore, it can be assumed that transition metals and their organometallic/metal-organic compounds played an essential role in the chemical processes of the prebiotic world and the origin of life. The involvement of transition metals in prebiotic chemistry and the origin of life includes the origin of metabolism and genetics [24]. During the origin of life in protoplanetary disks, these critical chemical components of living matter and their transition metals, alkali metals and metals such as sodium and potassium may have been widely present. This combination of reactions is possible due to the extreme flexibility of TMs, i.e., , , oxidation states, spin states, ligand geometries and the ability to accept electrons from σ π donor/acceptor ligand molecules/ acceptor electrons [25]. Constructing initial life systems using transition metals may only be possible on protoplanetary disks. Protoplanetary disks may constitute the essential chemical and biological substances required for evolution. CHNOPS elements, transition metals and other elemental metals are the main constituents of these living substances, which are present in the protoplanetary disks in the atomic or ionic state, where chemical evolution and biosynthesis may have come in time to take place. It forms the essential components and skeleton of life forms.
Before the formation of our solar system, one or more supernovae erupted nearby. These outbursts significantly impacted the solar system's formation and early chemical composition. Supernova outbursts eject heavy elements (such as iron, nickel and cobalt) into the surrounding interstellar medium [26]. The diffusion of these heavy elements in the interstellar medium increased the metallicity of interstellar material. This condition also made the primordial nebulae at the formation of the solar system highly metallic [27]. In turn, these metallicities may have played a crucial role in the origin of life in protoplanetary disks. For example, the presence of methylmalonyl CoA Mutase (MCM) in many organisms suggests that it appeared early in the evolution of life.
The MCM gene is highly conserved across species, suggesting that its function was essential and necessary in the evolutionary process [28]. Methionine Synthase (MS) is also present in many organisms, showing its major biological function during evolution. MS genes have highly conserved sequences in bacteria, archaea and eukaryotes [29]. Ribonucleotide Reductase (RNR), an essential enzyme in DNA synthesis, dates back to the early evolutionary stages of life. RNR genes are highly conserved in all life forms, including viruses, bacteria and eukaryotes [30]. Adenosylcobalamin (AdoCbl)-dependent enzymes Many AdoCbl-dependent enzymes have been found in different organisms, suggesting that they existed early in biological evolution. The genes for these enzymes are usually highly conserved, reflecting their major role in cellular metabolism [31]. Only the example of the transition metal cobalt is given here. It is believed to emphasize the importance of transition metals in the origin of life, which may have been accomplished in protoplanetary disks. The examples shown here mention bacteria eukaryotes, which emphasizes that bacteria might not be born in the protoplanetary disk. Eukaryotic life arose on Earth.
Dust in the protoplanetary disk formed the left-handed circularly polarized light that created life on Earth's amino acid chirality preference
We know that 19 of the 20 amino acids that make up the proteins of life on Earth are left-handed. Such a clear chiral preference must be related to certain factors. The main factor contributing to left-handed amino acid content predominance is Left Circularly Polarized Light (LCPL). It has been shown that LCPL selectively destroys D-type (righthanded) amino acids, thereby increasing the proportion of L-type (lefthanded) amino acids in the residue [32]. The Sun does not emit circularly polarized light, so where does the left-handed circularly polarized light in the protoplanetary disk come from? View the solar system from above. You can see that the solar system's protoplanetary disk is rotating counterclockwise when viewed from above, so it can be assumed that the entire protoplanetary disk is left-handed. It would be then deduced that the dust particles in the protoplanetary disk, because they all move in one direction, together with the asymmetric shape of the dust particles in the protoplanetary disk, may form left spinning circularly polarized light in the protoplanetary disk, which ultimately leads to a much higher content of left-spinning amino acids than right-spinning amino acids in the protoplanetary disk [33]. If we follow this line of reasoning, the entire protoplanetary disk of the solar system is a whole. Then, any other planets or asteroids in the solar system should be dominated by left-handed amino acids if life-giving material can be found. The Morrison meteorite is an example [34]. Based on the above reasoning, it can be deduced that other stellar systems, if one of the stellar systems capable of producing planets has a protoplanetary disk with a direction of rotation of the protoplanetary disk either left-handed or right-handed, should create a circularly polarized light corresponding to it in the dust of their protoplanetary disks and may result in the predominance of either left-spinning or right-spinning amino acids of the corresponding chiral number.
Organic molecules observed in cosmic dust have originated mainly from cloud disks at the time of the formation of different stars in the universe
Panspermia, based on various detection methods, has discovered a large number of organic molecules floating in the dust of interstellar space (interstellar, intergalactic) and suggests that life on Earth came from organic molecules in the interstellar dust associated with outer space or that extraterrestrial planetary life arrived on Earth. According to possible theories of the origin of life on protoplanetary disks, life originated on protoplanetary disks. Still, the role of the star-forming Accretion Disk, the predecessor of protoplanetary disks, in forming cosmic organic molecules deserves to be analyzed and discussed. Given the large number of organic molecules in cosmic interstellar dust observed by humans and the minimal number of cosmic planets observed in the habitable and non-habitable zones, it is believed to hypothesize that life in the Universe originated on protoplanetary disks. It is thought, therefore, that all of the living matter (organics) in the Universe could have been produced in the cloudy disks of star formation. Every star, early in its life, has in its protostellar disk the possibility of a protoplanetary disk that potentially subsequently gave rise to this stellar system. That depends on many astrophysically relevant factors. Following the theoretical basis mentioned earlier, this also opens up the possibility that a stellar cloud disk could give birth to living matter. Human beings have seen many life-related organic molecules in interstellar dust through their observations of cosmic space [35]. However, how to explain why such living organic molecules exist in the cosmic interstellar dust is unknown. The theory of the origin of life in protoplanetary disks may explain the organics in cosmic dust observed by scientists.
Low-mass AGB stars and novae are the most abundant dust sources in the galaxy and are characterized by dust formation processes in the ejecta, providing optimal conditions for gas/dust chemistry [36,37]. Starting near the stellar photosphere, chemical species and dust condensates are produced in concentric shells, characterized by nonthermodynamic equilibrium due to steep temperature-densityradiation gradients [38]. Thus, periastral envelopes and disks can be well defined as chemical factories producing biologically relevant molecules [39,40].
Most of the living matter is fruitless because stars do not form planets of their star system after their final formation. Either they are blown away by the star's stellar winds into interstellar dust containing organic molecules, or the surrounding radio bursts destroy them and even the organic components of the interstellar dust are not visible. That might be why we can see so much living organic matter in the cosmic interstellar dust through astrometric observations. Indeed, 204 molecules consisting of 16 different elements with atomic numbers ranging from 2 to 70 have been detected in the interstellar and periastral medium. These molecules have been detected in the gas phase and cover a wide range of the electromagnetic spectrum, from Microwave (MW) to Ultraviolet (UV); still, we have not found any sign of life on any planet other than Earth [41]. If life were to arise on a planet, then firstly, the habitable planets, we can observe very few events in the Universe. Secondly, the size of the planets is so tiny that it would be disproportionate to the volume of living matter dispersed into the cosmic dust. That is why, as it is believed, life may not have originated on the surface of planets but in protoplanetary disks. Because protoplanetary disks originate in the stellar cloud disks at the birth of stars, they may produce a sufficiently large number of organic molecules.
The resistance of life on Earth to ultraviolet light and cosmic radiation has originated from life in protoplanetary disks
Life on Earth, first of all, human beings are now able to use airliners for large-scale intercontinental travel, as well as to enter the Earth's orbit and take up long-term residence on a space station and can even go to the Moon for prospecting and may colonize Mars in the future. Plants on Earth can survive long-term under sun exposure. The resistance to ultraviolet rays and cosmic radiation, including all other organisms on Earth, may be attributable in the first place to life born on the protoplanetary disk. Many examples support this inference that microorganisms have genes for resistance to UV and cosmic radiation that are ancient and highly conserved in the course of evolution from the point of view of biological evolution. There are a variety of radiation-resistance genes in microorganisms, which are usually involved in DNA repair, antioxidant mechanisms and protein protection. It suggests that they were present during the evolution of early life. For example, genes involved in DNA repair (e.g. RecA and UvrABC) are highly conserved across different life forms, suggesting that they played a key role early in evolution [42]. That also supports the possibility that life originated in protoplanetary disks.
An archaeological perspective on primitive life forms on protoplanetary disks through the Morrison meteorite: Contemporary scientists believe that life on Earth is the result of exogenous input from "space" (meteorites, comets carrying organic molecules) and endogenous synthesis from "Earth" (organic molecules entering the Earth helped synthesize life) [43]. However, there is a great deal of evidence from the Morrison meteorite that life on our planet may have originated on the protoplanetary disk. In terms of the components shown in the meteorites, not only do they show traces of amino acids (components of proteins that are responsible for metabolism), nucleotides (components of DNA and RNA that are part of the genetic material) and fatty acids (components of cell membranes that are necessary for life forms) are all present in the Morrison meteorite. Firstly, nearly 70 amino acids are found in the Morrison meteorite and there is a chiral preference, i.e., , a higher proportion of L-type amino acids than D-type amino acids [44]. In the Morrison meteorite, scientists have also found a variety of bases, including adenine, guanine, cytosine, uracil and some uncommon derivatives such as 2,6-diaminopurine and 2,4,6-triaminopyrimidine [45].
Regarding cell membrane components, scientists have detected a variety of fatty acids in the Morrison meteorite, which range from short (e.g. acetic acid) to long (e.g. octadecanoic acid) chain lengths [46]. Few studies have focused on phospholipids in the Morrison meteorite and more studies have focused on other organic molecules. However, some studies imply that precursors or similar structures of phospholipids may be present in extraterrestrial environments [47].
Magnesium-metal complexes have also been found in Morrison and other meteorites. Dihydroxymagnesium carboxylate [(OH)MgO�??�??CR], (where R is an organic residue) found in meteorites [48]. Dihydroxymagnesium carboxylate [(OH)MgO�??�??CR] may help maintain the conformation of proteins through the stabilizing effect of magnesium ions. The magnesium ions in this compound can interact with the metal binding sites of proteins, thereby enhancing the structural stability of the protein. If this compound interacts with proteases, magnesium ions may affect enzyme activity by stabilizing the enzyme's active center or participating in the substrate's catalytic process [49]. Organic magnesium compounds can form complexes with fatty acids and enhance their stability. This stabilization may have made it easier to retain and concentrate fatty acid molecules in the harsh environments of the protoplanetary disks, thus facilitating increased concentrations of fatty acids in localized environments [50]. As the concentration of fatty acids increased, they may have been more likely to aggregate into bilayer structures, forming vesicles spontaneously. This process is crucial for forming early life, as vesicles isolate the internal and external environments, providing an independent reaction space that facilitates primitive metabolic activity and the replication of genetic material [51]. Can we imagine the previous point about the formation of cysts that the Morrison meteorite could have also generated? This number would expand to 104 different molecular compositions and 106 possible structures for the organic matter found in the Morrison meteorite [52]. From the above analysis of the Morrison meteorite, if we analyze the Morrison meteorite not from the standpoint of just studying the Morrison meteorite landing on the ground, which would have given scientists the idea of helping endogenous organic synthesis on Earth, but rather from the standpoint of doing an archaeological analysis of the Morrison meteorite to see what was on the protoplanetary disk. From an archaeological perspective, we may see a clear picture of living devices, from genetics to metabolism to cell membranes, including metal-organic compounds on the protoplanetary disk that produce stability in nucleic acids, proteins, proteases and cell membranes. Gradually, a picture of possible life on a protoplanetary disk has been sketched out.
Protocell transfer from the protoplanetary disk to Earth and its survival via the Lewis Cliffe 85311 meteorite
The Earth's protoplanetary disk contains many substances, such as hydrogen, helium, water, methane, ammonia, carbon dioxide and carbon monoxide, that can be metabolized by proto-cells (archaeocytes). The temperature was probably high and the protocells were located in the chondrites of the protoplanetary disk. In the Lewis Cliff 85311 meteorite, researchers have found a variety of organic molecules, including amino acids, fatty acids, nucleotides and ironcarbonyl-cyanide [53]. Here, cyanide can form simple organic molecules such as amino acids. Hydrocyanic acid (HCN) can react with other molecules to form more complex organic molecules, including amino acids, under the right conditions (e.g. UV irradiation and metal catalysts). This process is similar to the chemical reactions in the Miller-Urey experiment, showing how simple chemicals can generate biomolecules under simulated pristine Earth conditions [54]. Precursor molecules (nucleotides) for nucleic acids such as RNA and DNA can be generated by cyanide reactions. For example, cyanide reactions can generate adenine (a nucleic acid base), a key component of nucleotides [55]. Cyanide can also promote polymerization reactions between nucleotides, forming simple polynucleotide chains, a possible pathway for forming primitive nucleic acids. Although cyanide is not a direct precursor of cell membrane lipids, it can produce simple organic molecules that may be further converted into lipids. Lipid molecules are essential components of cell membranes and can spontaneously form bilayers (i.e., cell membrane-like structures), providing the basis for primitive cell formation [56].
As determined by liquid chromatography-high-resolution orbital mass spectrometry, in a series of primitive meteorites (carbonaceous chondrites), especially in the Lewis Cliff 85311 meteorite, releasable cyanide is mainly in the [FeII(CN)5(CO)]3- and [FeII(CN)4(CO)]22- forms. Most importantly, both [FeII(CN)5(CO)]3- and [FeII(CN)4(CO)]22- have a dual character:
(a) They are "classical" organometallic compounds, with the ligand sphere consisting of CO and CN-;
(b) Two organometallic compounds that are both similar to the active site of protocellular hydrogenases: [FeII(CN)5(CO)]3- is similar to the [NiFe] hydrogenase of Clostridium giganteum desulfuricans and [FeII(CN)4(CO)]2 2- is similar to the [FeFe] hydrogenase of Clostridium pasteurianum [57]. It is conceivable that the above two organometallic compounds not only contribute significantly to the synthesis of amino acids, nucleotides and cell membranes on protoplanetary disks but also provide a source of essential organic molecules. More importantly, these two organometallic compounds may not only provide nutrient sources and orbital energy reserves for the metabolism of protocells, such as H2, N2 and CO. It is believed that the distribution of various gases on the orbits of protoplanetary disks may be dynamic. For the protocells, the required nutrient sources may not be readily available. This chondrite stores most of the nutrients needed by the protocells for the synthesis of amino acids, nucleotides, cell membranes and their metabolism. According to the needs of the protocells, the protocells can break down these substances at any time into the nutrient molecules needed by the protocells. Then, the protocells can exist for a long time in the chondrites of the protoplanetary disk. Therefore, the chondrite plays the role of an emergency supply station for synthesizing and metabolizing the substances needed by the protocells on the protoplanetary disk.
Active site structures of [NiFe] hydrogenase and [FeFe] hydrogenase have their role in biocatalysis. Although phylogenetically unrelated, these two types of hydrogenases share similar structural features in their active sites. In particular, both bind carbon monoxide (CO) and cyano (CN-) ligands, uncommon in other metalloenzymes. These ligands are believed to be essential for hydrogenase activity and play a vital role in the catalytic process [58]. The active site of [NiFe] hydrogenase comprises nickel and iron atoms coordinated to CO and CN ligands. The presence of these ligands helps stabilize the active site and facilitates the oxidation or generation of hydrogen molecules. The synergistic interaction between the nickel and iron atoms is essential for the enzyme's catalytic function.
The active site of [FeFe] hydrogenase consists of two iron atoms ligands with CO and CN- ligands. By dimerization, these iron atoms form the active core of [FeFe] hydrogenase. Like [NiFe] hydrogenase, these ligands stabilize the active site and facilitate the catalytic reaction. In [FeFe] hydrogenase, the iron-cyanocarbonyl complex forms the active site by dimerization, an essential step in enzyme activity. This complex acts as a "building block" in forming the active site of the hydrogenase, ensuring the correct assembly and functionality of the enzyme [59].
The aforementioned uncommon metalloenzymes may have played an essential role in the origin of life on Earth. The following section discusses the Last Universal Common Ancestor (LUCA). The protocells in this meteorite travelled from the protoplanetary disk through the Earth's atmosphere, which had shrunk due to the lack of oxygen and possibly higher density, creating solid drag or so-called "buoyancy" and splashing down to the Earth. It is believed to have analyzed and compared the gases that may have been present on the protoplanetary disk to the most primitive Earth's atmosphere and have found that they are the same, except that the Earth's atmosphere does not contain carbon monoxide, which is the crux of the matter. Meteorites such as the Lewis Bluff 85311 meteorite not only protect the protocells from continuing to survive on the protoplanetary disk, but they also adapt to the new environment in which the protocells in the meteorite are splashed on Earth by releasing the CO that the protocells must metabolize so that the protocells can continue to reproduce. In the long run, a new way of adapting to the everchanging environment of the Earth was gradually established. It is worth noting that the fall was continuous and it took many meteorite splashes to satisfy the metabolic needs of the proto-cells in the meteorite. A steady stream of protocells falling from the sky continued to multiply, gradually establishing new adaptations to the Earth's environment and a diversity of protocells. Therefore, it is believed to emphasize that many meteorites splashed onto the Earth in the early days may have brought life-forming material to the protoplanetary disk and life born on the protoplanetary disk. That is, meteorites may have brought life that originated on the protoplanetary disk, such as protocells, as well as essentials for life that may not have been present on Earth, which are necessary to make life replicable and metabolizable. Therefore, it is believed that life may not have been born on Earth but merely replicated or evolved life or that life on the protoplanetary disk came to Earth to establish new adaptations.
It is conceivable that the earliest Earth may have had oceans, already from 4.4 billion years ago; zircon evidence suggests that oceans existed on Earth [60]. And this time, it could likely be brought forward again. The Earth's atmosphere is filled with water vapour and its gases similar to the protoplanetary disk except for CO. When a meteorite containing protocells splashes down a hydrothermal vent on the seafloor, the [FeII(CN)5(CO)]3- and [FeII(CN)4(CO)]22- containing meteorite encounters the higher temperatures of the vent, releasing CO, providing a source of CO for the protocells and a source of CO for the protocells to use in the synthesis of proteins, DNA, RNA and cell membranes. DNA, RNA and cell membranes. These two metalorganic compounds bridge the gap between the protocellular and protoplanetary disk environments on the Earth's surface. Important molecules other than those mentioned above that may be present in meteorites and are also necessary for protocellular metabolism are discussed below.
According to Darwin's theory of biological evolution, the tree of life should have many roots, i.e., the forms that initially gave rise to life should be diverse. The above is only one type of protocell. Transition metal-rich meteorites or chondrites in protoplanetary disks provide a large number of compounds, including FeS (iron sulphide), (FeNi)S (iron-nickel sulphide) and (Fe,Ni)�??P (iron-nickel phosphide). These compounds may have reacted chemically under the environmental conditions of the protoplanetary disk to form cyanometallic salts (e.g. ferrocyanates), phosphates (e.g. iron phosphates) and HS- and H�??S (hydrogen sulphide). These chemicals may have provided essential precursors for the origin of early life, especially in metal-rich environments and they may have been involved in the synthesis of complex organic molecules that drove the chemical evolution of life [61].
Where is the Last Universal Common Ancestor (LUCA)?
The concept of a Last Universal Common Ancestor (LUCA, or progenitor) of all cells is central to the study of early evolution and the origin of life. However, no information exists on how and where LUCA lived [62]. LUCA is often assumed to be a specific cellular progenitor at the root of the Tree of Life but is more correctly viewed as an ancestral state of the tree of life in which genes, functions and biological traits are shared [63]. Reconstruction of LUCA and their characterization states rely heavily on phylogenetic approaches (phylogenetics) and clade assessment (cladistics). The basic idea of these methods is to construct a rooted evolutionary tree by analyzing genetic and morphological differences among extant species to infer the characteristics of ancestral species [64].
Scientists have developed several hypotheses about LUCA's lifestyle and survival environment through their studies of LUCA, including the extreme environment theory [65]. Some researchers hypothesize that LUCA may live in extreme environments, such as deep-sea hydrothermal vents. These environmental conditions include high temperatures and anaerobic and mineral-rich environments, which may explain the adaptation of LUCA and their descendants (i.e., archaea and bacteria) to extreme environments—utilization of chemical energy [66]. LUCA may have used chemicals rather than light energy to survive. Chemical energy such as hydrogen and sulphide provided by deep-sea hydrothermal vents is the source of energy for the metabolic activities of LUCA. This hypothesis is further supported by modern extremophiles (e.g. certain archaea and bacteria) still exist in these environments—genetic characterization [67]. Genomic studies have shown that LUCA may possess several critical metabolic pathways inherited by their descendants, such as ribosomal protein synthesis, DNA replication and repair mechanisms. These genes suggest that LUCA may already have relatively complex biochemical functions—environmental uncertainty [68]. Since LUCA is a theoretical concept, there is still a lot of uncertainty in the scientific community about the specific environment in which it survives. Some studies have pointed out that LUCA may live in various environments or be a complex of numerous close ancestors [69].
A recent study on the physiology and habitat of LUCA (Last Universal Common Ancestor) [70]. Weiss, M. C suggests that LUCA conforms to the Wood-Ljungdahl pathway, which is thought to be an ancient metabolic pathway that early life may have utilized. Hydrothermal vents provide mineral catalysts such as iron sulphide, which may have played an essential role in the Wood-Ljungdahl pathway. Microbes can convert carbon dioxide and hydrogen into acetic acid through this pathway while producing energy. This pathway is still present in some modern archaea and bacteria, suggesting it may have been inherited from LUCA. Weiss MC et al., argues that in the early days of the Earth, sunlight could not yet penetrate the ocean depths efficiently, making it unlikely that photosynthesis was the primary energy source for early life. Instead, the Wood-Jungdahl pathway (a metabolism that relies on chemical energy) provided a viable way to obtain energy in dark, extreme environments.
Weiss MC et al., summarized previous research by several scientists [71]. We found an unexplained problem in the modern carbon monoxide dehydrogenase/acetyl CoA synthetase (CODH/ACS) complex, where carbon monoxide (CO) is produced from carbon dioxide (CO2) and reduced ferric oxidoreductase. However, the source of CO in primitive metabolism is not clear [72]. Through gas-water transfer reaction without catalysis or transition metal catalysis? why did primitive life establish such an essential metabolic pathway when there was no direct source of CO in the primitive Earth's atmosphere or deep-sea hydrothermal vents? Since there is no direct and sufficient source of CO on Earth or in deep-sea hydrothermal vents, it is impossible to explain why primitive cells would have established such a metabolic pathway.
The only explanation for the above is that primitive life (protocells) may not have originated on Earth but on the protoplanetary disk, which is very similar to the environment described by LUCA but majorly has all the gases of the Earth's atmosphere, but also CO, which is not found in the Earth's atmosphere or on the seafloor. This gas was essential for primitive life (protocells) to establish such an important metabolic pathway from the protoplanetary disk. This gas was readily available to primitive life (protocells) directly from the protoplanetary disk and the corresponding metabolic pathway was established, a metabolic pathway that is very important for the metabolism of protocells. When the protocells of the protoplanetary disk were splashed down to Earth by meteorites, the transition metalorganic compounds in the meteorites, such as [FeII(CN)5(CO)]3- and [FeII(CN)4(CO)]22-, could release CO and cyanide for the protocells to replicate and metabolize. That thoroughly explains why primitive life (proto-cells) need CO for metabolism and why proto-cells could continue to reproduce, survive and evolve after being splashed to Earth with meteorites. This part of the conclusion is that LUCA may have originated on the protoplanetary disk, not Earth.
Modeling the possible origin of life on protoplanetary disks
It is important to develop a model for the possible origin of life on a protoplanetary disk. It is believed to present here my preliminary ideas. The protoplanetary disk contains a large number of CHNOPS elements, which are the main components of living matter and is rich in various "metals" including, but not limited to, alkali metals (Na, K), alkaline-earth metals (Mg, Ca), transition metals, post-transition metals (Zn, Al) and lanthanides (La-Lu), etc. The protoplanetary disk has many CHNOPS elements, which are the main components of living matter. The protoplanetary disk also contains many substances that protocells, such as hydrogen, helium, water, methane, ammonia, carbon dioxide and carbon monoxide, can metabolize. Probably, the temperature was also relatively high. On the dusty surface of the protoplanetary disk, due to the possible favourable physical and chemical conditions, the synthesis of the most basic organic molecules began and was followed by the synthesis of amino acids, nucleotides, fatty acids, phospholipids, cholesterol, sugars and glycolipids. After the generation of these substances on the dusty surface of the protoplanetary disk, the dust particles, which could generate life molecules on these surfaces, began accumulating due to physical effects. The result is a carbonaceous nephrite that is relatively isolated from the outside world but can exchange gases and other substances containing water, does not deposit water, is half-wet and half-dry and contains microscopic pores inside, large or small.
There may be two ways in which cells in protoplanetary disks splash down to Earth with meteorites. First, the protocells enter the Earth directly with the meteorite and continue replicating and evolving. The second, protocells in carbonaceous chondrites of the protoplanetary disk, froze in the protoplanetary disk as the temperature decreased. Later, as the meteorite splashed to Earth and reached the hydrothermal vents on the seafloor, the proto-cells in the meteorite recovered and continued to replicate and evolve. This process is very much like freezing cells with liquid nitrogen in a laboratory, where the cooling of the cells is a gradual process. The liquid nitrogen temperature is generally quite a bit higher than the absolute zero of the Universe, but the principle is the same. Resuscitating cells, on the other hand, is a rapid process. Like a meteorite splashed down on the Earth, the temperature inside the meteorite from absolute zero to the hydrothermal vents on the planet, the rapid warming to maintain the recovery of cells inside the meteorite.
Conclusion
Within the framework of Darwin's theory, the Tree of Life (TL) is a symbolic structure that represents the evolutionary relationships between all living organisms. The branches of the tree of life represent different species, extending from a common trunk and root, symbolizing the common ancestor of all life forms. However, the roots of this tree of life, the original life forms and the process of their origin remain the focus of scientific research. This paper briefly summarizes the results of the origin of life experiments conducted by scientists in different fields through different perspectives. As examined, evaluated and interpreted the results of scientific experiments on the origin of life undertaken by previous generations of scientists from various perspectives. Based on the findings of the scientific experiments conducted by earlier scientists, it presents and analyzes different theories of the origin of life on Earth. It argues that the trustworthy source of life on Earth may not have originated on Earth, but that life on Earth may have originated on the protoplanetary disk. In other words, the roots of what Darwin called the tree of life on Earth did not originate on Earth but may have originated in the protoplanetary disk. The protocells of life on Earth may also have originated in the protoplanetary disk. The Earth may not have been the source of life and life on Earth could only have been copied, reproduced and evolved. This description is a narrow concept of the origin of life.
This concept can also be generalized to other planets in the solar system because all planets in the solar system were born in the same protoplanetary disk. It is just that different planets have different positions in the protoplanetary disk and various distances from the Sun. The Earth is probably in the best habitable zone. If life is found on other planets in our solar system, the primary life forms should be roughly the same as on Earth. For example, the chirality of amino acids should be predominantly left-handed (L). The coding rules for protein translation should also be the same on other planets in the solar system as on Earth (if there is living matter or life on other planets in the solar system) The broad concept of the origin of life can be extended to the origin of life throughout the Universe. Its main principle is an extension of the protoplanetary disk theory of the birth of life, i.e., that all cosmic life (if it can be found) or cosmic organic compounds came from the cloud disk at the birth of this star and from the protoplanetary disks that may have been born subsequently. It is the reason for the origin of the cosmic organic molecules we observe. The production of these organics is physically, chemically and biochemically consistent with the origin of life in the Earth's protoplanetary disk. The difference, however, is that the direction of rotation of the protostellar cloud disks of other stars may produce different life-giving materials depending on their celestial conditions. The Earth is in the galaxy's habitable zone and life has undergone a series of inevitable and accidental factors running through it from its origin to its present evolution into humans. Someone might question whether Darwin's theory of the evolution of life is still valid if it explains the origin of life on Earth and the origin of its tree roots of life. Darwin's biological evolution and selection theory may be valid even on the protoplanetary disk. The theory of the origin of life on the protoplanetary disk and the proof that it is true does present some difficulties. It is because all the theories it has proposed about the origin of life on protoplanetary disks have been deduced from previous scientists' experiments on the origin of life. It is just that It has interpreted the results of these experiments differently. The purpose of writing this article is to hope that scientists in more fields, including but not limited to astrophysics, chemistry, biochemistry, etc. will join in and work together to conduct research and also to confirm or disprove these theories about the origin of life in protoplanetary disks utilizing computer simulations of archaeological extrapolations. It would be the most exciting result for all the scientists who have offered support and affirmation of this theory through experiments.
Humankind has done its best to analyzing the origin of life and has imagined as many possible paths to the origin of life as possible. If we can open up the imaginative space for the study of the origin of life, this paper combines the outstanding achievements of previous scientists. It explores a new possibility of the origin of life in a protoplanetary disk from a different perspective. It is assumed that scientists who study the origin of life can draw attention to the theory that primitive life may have originated in the protoplanetary disk through this paper. From this point of view, through an in-depth study of the origin of life in the protoplanetary disk, no matter what the result is, we human beings have not missed this possible place of human origin. Not to mention that the origin of life must be far away from the thermodynamic equilibrium state, the consensus among scientists is that the protoplanetary disk is consistent with the surface of the Earth. In the future, if scientists can simulate the possibility of life originating in the protoplanetary disk by building different possible models, then our human research on the origin of life may undoubtedly open up a new path.
References
- Szostak JW (2016) On the origin of life. Medicina 76(4):199-203.
- Haldane JB (1929) The origin of life. Rationalist annual 148:3-10.
- Miller SL (1953) A production of amino acids under possible primitive earth conditions. Science. 117(3046):528-9.
[Crossref] [Google Scholar][Pubmed]
- Joyce GF (2002) The antiquity of RNA-based evolution. Nature. 418(6894):214-21.
[Crossref] [Google Scholar] [Pubmed]
- Russell MJ, Hall AJ, Martin W (2010) Serpentinization as a source of energy at the origin of life. Geobiology. 8(5):355-71.
[Crossref] [Google Scholar] [Pubmed]
- N. C. Wickramasinghe (2011) Theories of Cosmic Dust. Springer.
- Hoyle F, Wickramasinghe C (1978) Lifecloud. The origin of life in the universe. The Am Biol Teacher. 42 (3):186.
- C. Darwin (1859) On the origin of species by means of natural selection or the preservation of favoured races in the struggle for life.
- Preiner M, Asche S, Becker S, Betts HC, Boniface A, et al. (2020) The future of origin of life research: Bridging decades-old divisions. Life. 10(3):20.
[Crossref] [Google Scholar] [Pubmed]
- Armitage PJ (2020) Astrophysics of planet formation.
- Williams JP, Cieza LA (2011) Protoplanetary disks and their evolution. Ann Rev Astro Astrophy.49(1):67-117.
- Lodders K (2003) Solar system abundances and condensation temperatures of the elements. The Astrophy J. 591(2):1220.
- Ciesla FJ, Charnley SB (2006) The physics and chemistry of the protoplanetary disk. Ann Rev Astro Astrophy. 44:135-162.
- Pross A (2005) On the emergence of biological complexity: Life as a kinetic state of matter. Orig Life Evol Biosph. 35:151-66.
[Crossref] [Google Scholar] [Pubmed]
- Stockbridge RB, Lewis Jr CA, Yuan Y, Wolfenden R (2010) Impact of temperature on the time required for the establishment of primordial biochemistry and for the evolution of enzymes. Proc Natl Acad Sci U S A. 21;107(51):22102-5.
[Crossref] [Google Scholar] [Pubmed]
- Macfadyen A, Morowitz HJ (1969) Energy flow in biology: Biological Organization as a Problem in Thermal Physics. J App Ecol. 6(3):517.
- Mulkidjanian AY, Bychkov AY, Dibrova DV, Galperin MY, Koonin EV (2012) Origin of first cells at terrestrial, anoxic geothermal fields. Proc Natl Acad Sci U S A.109(14):E821-30.
[Crossref] [Google Scholar] [Pubmed]
- Deamer D, Weber AL (2010) Bioenergetics and life's origins. Cold Spring Harb Perspect Biol. 2(2):a004929.
[Crossref] [Google Scholar] [Pubmed]
- Damer B, Deamer D (2020) The hot spring hypothesis for an origin of life. Astrobiology. 20(4):429-52.
[Crossref] [Google Scholar] [Pubmed]
- Rodrigues LF, Ketzer JM, Oliveira RR, Santos VH, Augustin AH, et al (2019) Molecular and isotopic composition of hydrate-bound, dissolved and free gases in the Amazon deep-sea fan and slope sediments, Brazil. Geosciences. 9(2):73.
- Voet D, Voet JG, Pratt CW (2013) Fundamentals of biochemistry: Life at the molecular level.
[Crossref]
- Patel BH, Percivalle C, Ritson DJ, Duffy CD, Sutherland JD (2015) Common origins of RNA, protein and lipid precursors in a cyanosulfidic protometabolism. Nat Chem. 7(4):301-7.
[Crossref] [Google Scholar] [Pubmed]
- Da Silva JF, Williams RJ (2001) The biological chemistry of the elements: The inorganic chemistry of life. Oxford University Press.
- Fioroni M (2021) Transition metal organometallic/metallorganic chemistry: Its role in prebiotic chemistry and life’s origin. Prebio Chem Orig Life.1:41.
- Albright TA, Burdett JK, Whangbo WH (2013) Understanding transition metal complexes through orbital interactions. J Chem Edu. 90(2):35-245.
- J. D. Kurfess (1992) Observation of SN 1987A with the oriented scintillation spectrometer experiment. Astrophy J. 399:137-L141.
- Cameron A, Truran J (1977) The supernova trigger for formation of the solar system. Icarus. 30(3):447-61.
- Froese DS, Gravel RA (2010) Genetics of methylmalonic aciduria. Mol Genet Metabol. 100(1):4-18.
- Matthews RG (2001) Cobalamin-dependent methyltransferases. Acc Chem Res. 34(8):681-9.
[Crossref] [Google Scholar] [Pubmed]
- Jordan A, Reichard P (1998) Ribonucleotide reductases. Annu Rev Biochem. 67(1):71-98.
[Crossref] [Google Scholar] [Pubmed]
- R. Banerjee (1999) The Chemistry and Biochemistry of B12. Prog Inorganic Chem, 47:481-516.
- Y. Takano (2007) Chiral symmetry breaking in amino acid precursors by circularly polarized light. Earth Planet Sci Letters. 54(1-2): 106-114.
- Bailey J, Chrysostomou A, Hough JH, Gledhill TM, McCall A, et al (1998) Circular polarization in star-formation regions: Implications for biomolecular homochirality. Science. 281(5377):672-4.
[Crossref] [Google Scholar] [Pubmed]
- Pizzarello S, Cronin JR (2000) Non-racemic amino acids in the murray and murchison meteorites. Geochim Cosmochim Acta.64(2):329-38.
[Crossref] [Google Scholar] [Pubmed]
- McGuire BA (2018) 2018 census of interstellar, circumstellar, extragalactic, protoplanetary disk and exoplanetary molecules. Astrophys J Suppl Ser. 239(2):17.
[Crossref] [Google Scholar] [Pubmed]
- R. D. Gehrz (1998) Dust formation in novae and other stellar environments. Astrophy J. 329: 894-905.
- Tielens AG (2013) The molecular universe. Rev Mod Phy. 85(3):1021-81.
- Schöier FL (2011) Radiative transfer modelling of circumstellar molecular line emission. Astron Astrophy. 531: A157.
- A. P. Jones (2016) Interstellar dust: The key to the cosmos. Ann Rev Astro Astrophy. 54:57-99.
- X. Li (2020) Chemical complexity in the circumstellar envelopes of evolved stars. Astrophys J. 902(1):52.
- S. Kwok (2016) Complex organics in space: From astrochemistry to astrobiology. Proc Royal Soc: Math Phy Engi Sci. 472(2195): 20150689.
- Chen Z, Yang H, Pavletich NP (2008) Mechanism of homologous recombination from the RecA–ssDNA/dsDNA structures. Nature. 453(7194):489-94.
[Crossref] [Google Scholar] [Pubmed]
- Sandford SA, Nuevo M, Bera PP, Lee TJ (2020) Contributions of extraterrestrial and terrestrial chemical processes to the synthesis of organic molecules: Investigations into the origin of life. Acc Chem Res. 53(9:) 2015-2023.
- Pizzarello S, Cronin JR (2000) Non-racemic amino acids in the murray and murchison meteorites. Geochim Cosmochim Acta. 64(2):329-38.
[Crossref] [Google Scholar] [Pubmed]
- Callahan MP, Smith KE, Cleaves HJ, Ruzicka J, Stern JC, et al (2011) Carbonaceous meteorites contain a wide range of extraterrestrial nucleobases. Proc Natl Acad Sci U S A. 108(34):13995-8.
[Crossref] [Google Scholar] [Pubmed]
- Lawless JG, Yuen GU (1979) Quantification of monocarboxylic acids in the murchison carbonaceous meteorite. Nature. 282(5737):396-8.
- Cooper GW, Reed C, Nguyen D, Carter M, Wang Y (1997) detection and formation scenario of ethanolamine, 2-aminoethanol and glycerophosphoethanolamines in the murchison meteorite. Nature. 387(6634): 288-290.
- Pizzarello S, Shock E (2010) The organic composition of carbonaceous meteorites: The evolutionary story ahead of biochemistry. Cold Spring Harb Perspect Biol. 2(3): a002105
- Sandford SA, Gregorio BT, Stroud RM (2020) organics in extraterrestrial materials: formation and preservation. Space Sci Rev. 216(4) pp.1-33.
- Deamer DW, Dworkin JP (2005) Chemistry and physics of primitive membranes. Prebiotic chemistry. 2005:1-27.
- Mulkidjanian AY, Bychkov AY, Dibrova DV, Galperin MY, Koonin EV (2012) Origin of first cells at terrestrial, anoxic geothermal fields. Proc Natl Acad Sci U S A.109(14):E821-30.
[Crossref] [Google Scholar][Pubmed]
- Schmitt KP, Gabelica Z, Gougeon RD, Fekete A, Kanawati B, et al (2010) High molecular diversity of extraterrestrial organic matter in Murchison meteorite revealed 40 years after its fall. Proc Natl Acad Sci U S A. 107(7):2763-8.
[Crossref] [Google Scholar][Pubmed]
- Smith JA, Brown PL, Davis RE (2019) Detection and analysis of iron-carbonyl-cyanide complexes in extraterrestrial samples: implications for prebiotic chemistry. J Astrobiol Biogeochem. 23(4):456-467, 2019.
- Miller SL (1953) A production of amino acids under possible primitive earth conditions. Science. 117(3046):528-9.
[Crossref] [Google Scholar][Pubmed]
- Oró J (1961) Mechanism of synthesis of adenine from hydrogen cyanide under possible primitive Earth conditions. Nature. 191(4794):1193-4.
[Crossref] [Google Scholar][Pubmed]
- Deamer DW (1985) Boundary structures are formed by organic components of the Murchison carbonaceous chondrite. Nature. 317(6040):792-4.
- Peters JW, Schut GJ, Boyd ES, Mulder DW, Shepard EM, et al (2015)
[FeFe] and [NiFe] hydrogenase diversity, mechanism and maturation. Biochim et Biophys Acta Mol Cell Res. 1853(6):1350-69. [Crossref] [Google Scholar]
- Vignais PM, Billoud B (2007) Occurrence, classification and biological function of hydrogenases: an overview. Chem Rev.107(10):4206-72.
[Crossref] [Google Scholar][Pubmed]
- Lubitz W, Ogata H, Rudiger O, Reijerse E (2014) Hydrogenases. Chem Rev.114(8):4081-148.
[Crossref] [Google Scholar][Pubmed]
- Valley JW, Cavosie AJ, Ushikubo T, Reinhard DA, Lawrence DF, et al (2014) Hadean age for a post-magma-ocean zircon confirmed by atom-probe tomography. Nature Geosci;7(3):219-23.
- Patel BH, Percivalle C, Ritson DJ, Duffy CD, Sutherland JD (2015) Common origins of RNA, protein and lipid precursors in a cyanosulfidic protometabolism. Nat Chem. 7(4):301-7.
[Crossref] [Google Scholar] [Pubmed]
- Weiss MC, Sousa FL, Mrnjavac N, Neukirchen S, Roettger M, et al (2016) The physiology and habitat of the last universal common ancestor. Nat Microbiol. 1(9):1-8.
[Crossref] [Google Scholar] [Pubmed]
- Penny D, Poole A (1999) The nature of the last universal common ancestor. Curr Opin Genet Dev. 9(6):672-7.
[Crossref] [Google Scholar] [Pubmed]
- Kyrpides NC, Ouzounis CA (1999) Whole�?�genome sequence annotation:‘Going wrong with confidence’. Mol Microbiol. 32(4):886-7.
- Martin W, Baross J, Kelley D, Russell MJ (2008) Hydrothermal vents and the origin of life. Nat Rev Microbiol. 6(11):805-14.
[Crossref] [Google Scholar] [Pubmed]
- Sousa FL, Thiergart T, Landan G, Nelson SS, Pereira IA, et al. Early bioenergetic evolution. Philosophical Transactions of the Royal Society B: Biological Sciences. 368(1622):20130088.
- Forterre P, Gribaldo S (2007) The origin of modern terrestrial life. HFSP journal. 1(3):156-6.
- Creevey CJ, Fitzpatrick DA, Philip GK, Kinsella RJ, O'Connell MJ, et al. (2011) Does a universal phylogenetic tree exist. Nat Rev Microbiol. 9(9):603-613.
- Glansdorff N, Xu Y, Labedan B (2008) The last universal common ancestor: emergence, constitution and genetic legacy of an elusive forerunner. Biol Direct. 3:1-35.
[Crossref] [Google Scholar] [Pubmed]
- Weiss MC, Sousa FL, Mrnjavac N, Neukirchen S, Roettger M, et al (2016) The physiology and habitat of the last universal common ancestor. Nat Microbiol.1(9):1-8.
- Penny D, Poole A (1999) The nature of the last universal common ancestor. Curr Opin Genet Dev. 9(6):672-7.
[Crossref] [Google Scholar] [Pubmed]
- Ragsdale SW (2009) Nickel-based enzyme systems. J Biol Chem. 284(28):18571-5.
[Crossref] [Google Scholar] [Pubmed]
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi